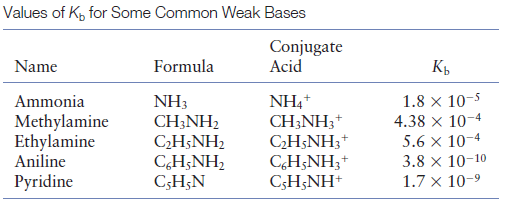
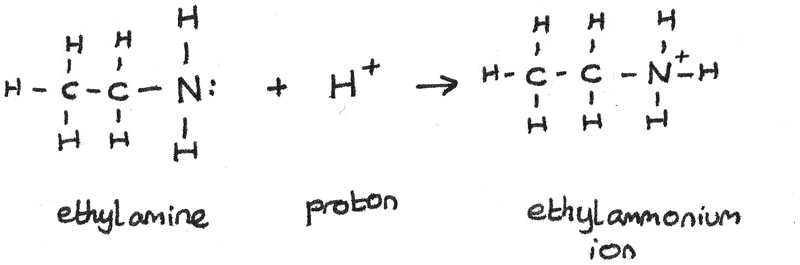
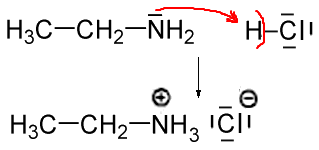
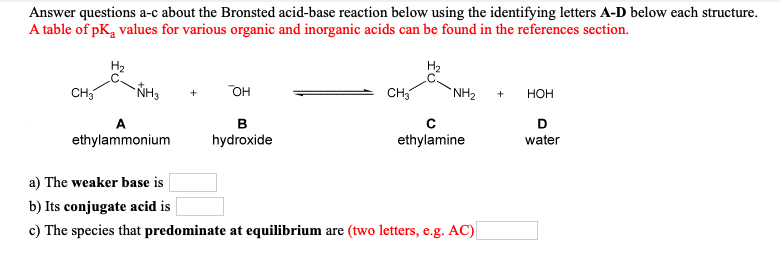
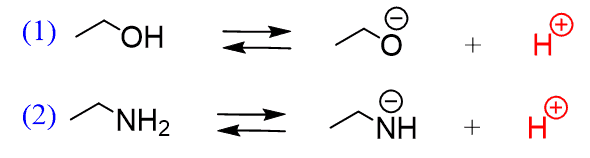
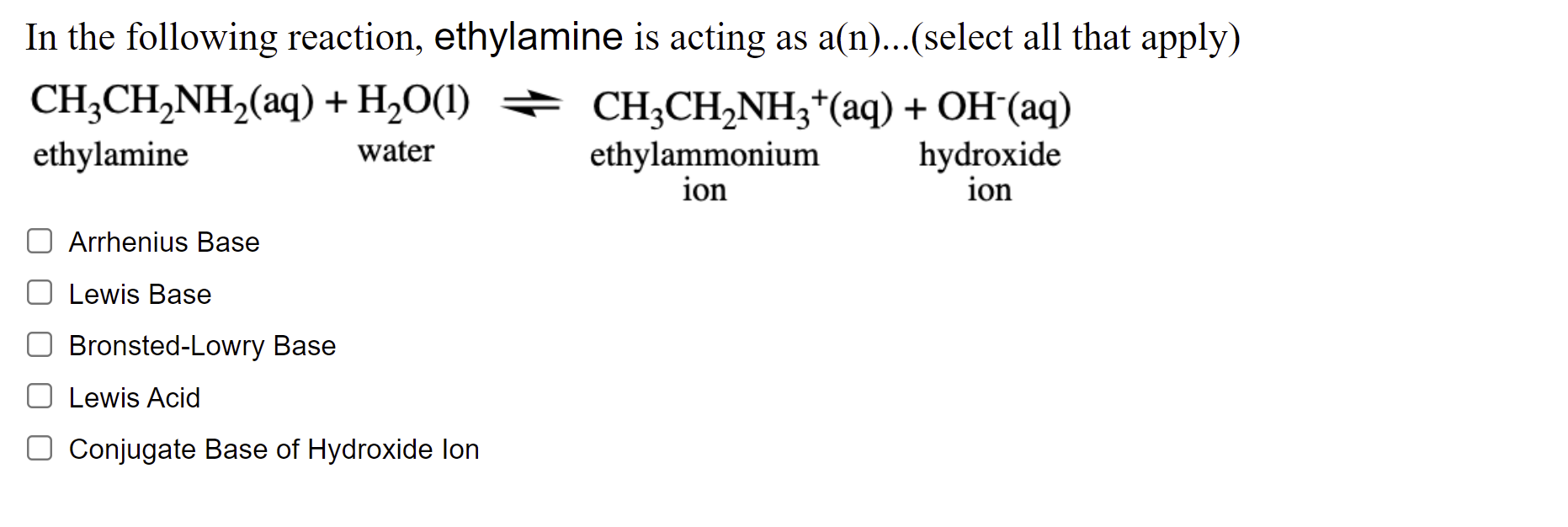
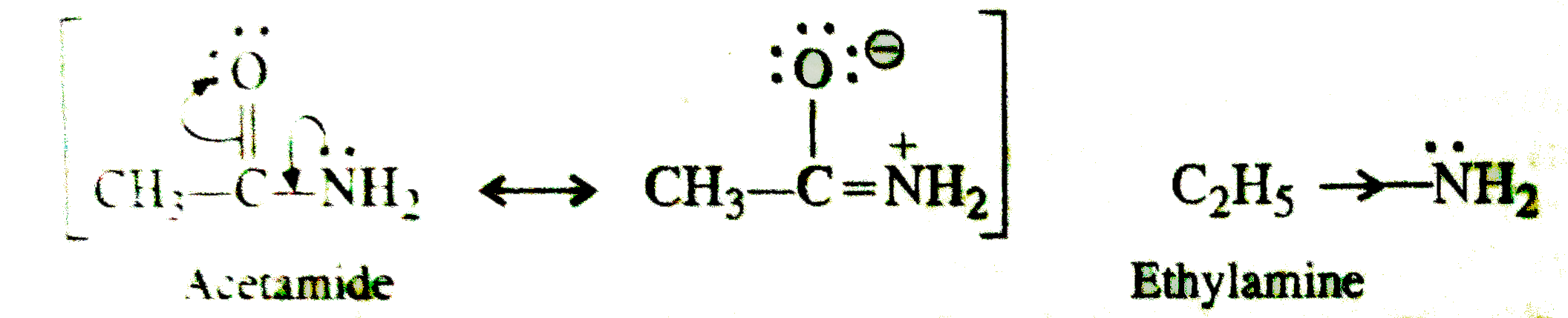Acid-base chemistry of aliphatic amines weak bases pKb Kb values why stronger than aomatic amines reactions with acids primary secondary tertiary balanced neutralisation equations organic nitrogen compounds organonitrogen molecules advanced A level

organic chemistry - Is Bromide anion acting as a base in reaction with ethylamine and methylbromide? - Chemistry Stack Exchange
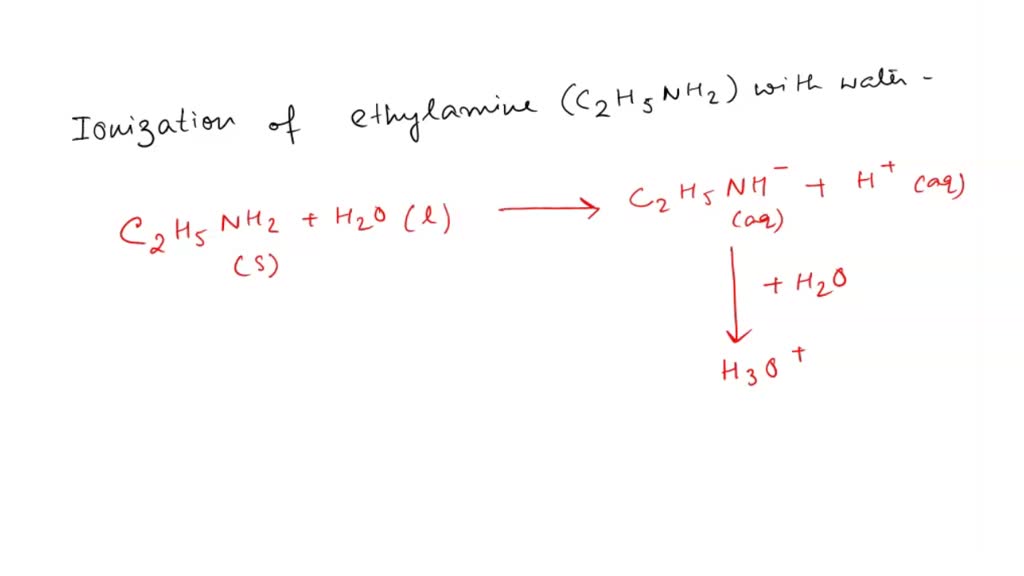
SOLVED: Write the equation for the ionization of ethylamine (C2H5NH2), a weak molecular base, with water
Ethylamine organic base molecule. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles: hydrogen (white), carbon (grey), nitrogen (blue Stock Photo - Alamy
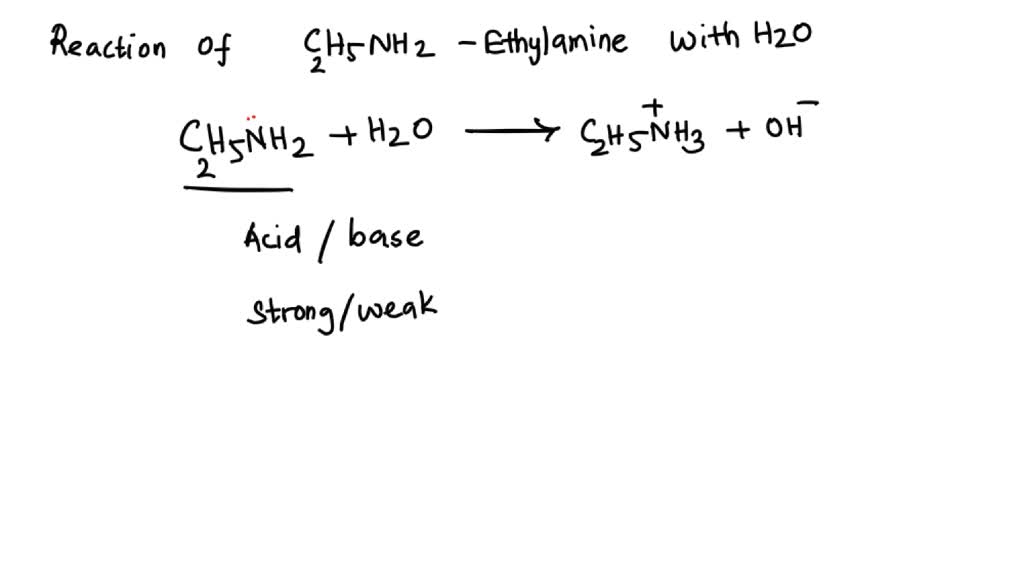
SOLVED: 5. When added to water, ethylamine undergoes the following reaction: C2H5NH2 + H2O C2H5NH3+ + OH- Is ethylamine an acid or a base in this reaction? Strong or weak? How do